购物车
 您的购物车当前为空
您的购物车当前为空
Primidone (NCI-C56360) 是一种强效抗惊厥试剂。它是神经元性电压门控钠通道阻滞剂,在癫痫、原发性震颤和精神疾病的研究中具有价值。
别名 去氧苯比妥, 扑米酮, Primaclone, NCI-C56360, Mysoline
Primidone (NCI-C56360) 是一种强效抗惊厥试剂。它是神经元性电压门控钠通道阻滞剂,在癫痫、原发性震颤和精神疾病的研究中具有价值。
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25 mg | ¥ 148 | 现货 | |
| 50 mg | ¥ 198 | 现货 | |
| 100 mg | ¥ 328 | 现货 | |
| 500 mg | ¥ 783 | 现货 | |
| 1 g | ¥ 1,150 | 现货 | |
| 1 mL x 10 mM (in DMSO) | ¥ 166 | 现货 |
TargetMol的所有产品仅用作科学研究或药证申报,不能被用于人体,我们不向个人提供产品和服务。请您遵守承诺用途,不得违反法律法规规定用于任何其他用途。

| 产品描述 | Primidone (NCI-C56360) is a potent anticonvulsant agent. It is a neuronal voltage-gated sodium channel blocker and has value in the study of epilepsy, essential tremor, and psychiatric disorders. |
| 别名 | 去氧苯比妥, 扑米酮, Primaclone, NCI-C56360, Mysoline |
| 分子量 | 218.25 |
| 分子式 | C12H14N2O2 |
| CAS No. | 125-33-7 |
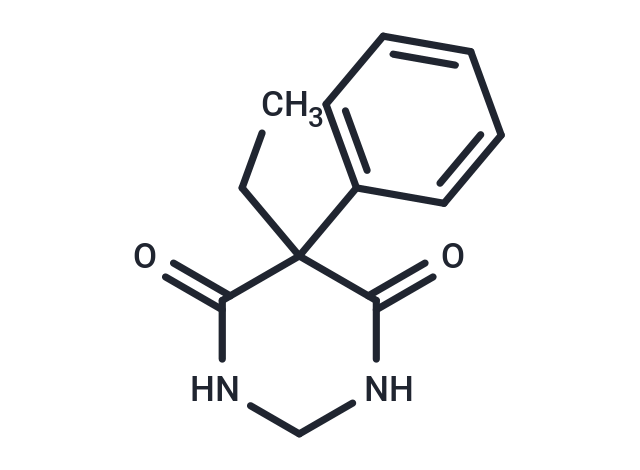
| Smiles | CCC1(C(=O)NCNC1=O)C1=CC=CC=C1 |
| 密度 | 1.138g/cm3 |
| 存储 | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. 实际储存温度请以COA为准 | |||||||||||||||||||||||||||||||||||
| 溶解度信息 | DMSO: 70 mg/mL (320.73 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
| 体内实验配方 | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 2 mg/mL (9.16 mM), Sonication is recommended. 请按顺序添加溶剂,在添加下一种溶剂之前,尽可能使溶液澄清。如有必要,可通过加热、超声、涡旋处理进行溶解。工作液建议现配现用。以上配方仅供参考,体内配方并不是绝对的,请根据不同情况进行调整。 | |||||||||||||||||||||||||||||||||||
溶液配制表 | ||||||||||||||||||||||||||||||||||||
DMSO
该溶液配制表仅适用于固体产品。对于液体产品,请根据标明的浓度或密度计算稀释方案。 | ||||||||||||||||||||||||||||||||||||
对于不同动物的给药剂量换算,您也可以参考 更多