购物车
 您的购物车当前为空
您的购物车当前为空
Midodrine ((±)-Midodrin) 是α1受体激动剂,用于研究家族性自主神经异常以及直立性低血压。
别名 米多君, (±)-Midodrin
Midodrine ((±)-Midodrin) 是α1受体激动剂,用于研究家族性自主神经异常以及直立性低血压。
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1 mg | ¥ 185 | 现货 | |
| 2 mg | ¥ 258 | 现货 | |
| 5 mg | ¥ 413 | 现货 | |
| 10 mg | ¥ 663 | 现货 | |
| 25 mg | ¥ 1,080 | 现货 | |
| 50 mg | ¥ 1,630 | 现货 | |
| 100 mg | ¥ 2,430 | 现货 | |
| 1 mL x 10 mM (in DMSO) | ¥ 397 | 现货 |
TargetMol的所有产品仅用作科学研究或药证申报,不能被用于人体,我们不向个人提供产品和服务。请您遵守承诺用途,不得违反法律法规规定用于任何其他用途。
该分子属于定制产品。TargetMol拥有优秀的合成团队,经验和能力,可以为您提供高性价比的产品。 如您有任何问题,欢迎咨询,我们将竭诚为您服务。

| 产品描述 | Midodrine ((±)-Midodrin), an adrenergic alpha-1 agonist, belongs to the class of medications known as vasopressors. |
| 别名 | 米多君, (±)-Midodrin |
| 分子量 | 254.28 |
| 分子式 | C12H18N2O4 |
| CAS No. | 42794-76-3 |
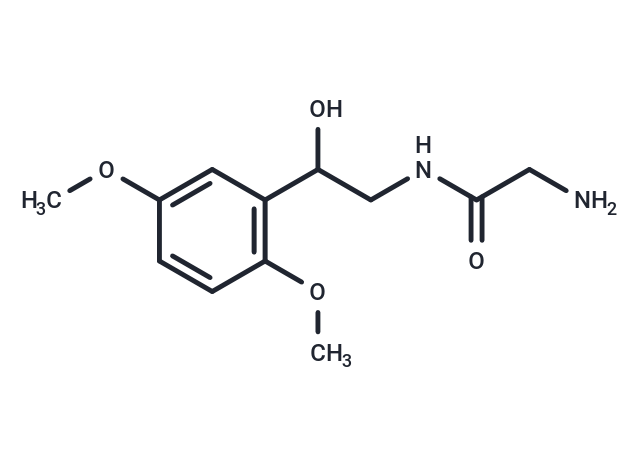
| Smiles | C(CNC(CN)=O)(O)C1=C(OC)C=CC(OC)=C1 |
| 密度 | 1.204 g/cm3 (Predicted) |
| 存储 | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. 实际储存温度请以COA为准 | |||||||||||||||||||||||||||||||||||
| 溶解度信息 | DMSO: 50 mg/mL (196.63 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
溶液配制表 | ||||||||||||||||||||||||||||||||||||
DMSO
该溶液配制表仅适用于固体产品。对于液体产品,请根据标明的浓度或密度计算稀释方案。 | ||||||||||||||||||||||||||||||||||||
对于不同动物的给药剂量换算,您也可以参考 更多