购物车
 您的购物车当前为空
您的购物车当前为空
Famciclovir (BRL 42810) 是一种单纯疱疹病毒核苷类似物 DNA 聚合酶抑制剂。
别名 泛昔洛韦, BRL 42810
Famciclovir (BRL 42810) 是一种单纯疱疹病毒核苷类似物 DNA 聚合酶抑制剂。
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25 mg | ¥ 289 | 现货 | |
| 50 mg | ¥ 413 | 现货 | |
| 100 mg | ¥ 690 | 现货 | |
| 200 mg | ¥ 1,090 | 现货 | |
| 500 mg | ¥ 2,120 | 现货 | |
| 1 mL x 10 mM (in DMSO) | ¥ 228 | 现货 |
TargetMol的所有产品仅用作科学研究或药证申报,不能被用于人体,我们不向个人提供产品和服务。请您遵守承诺用途,不得违反法律法规规定用于任何其他用途。

| 产品描述 | Famciclovir (BRL 42810) is a Herpes Simplex Virus Nucleoside Analog DNA Polymerase Inhibitor. The mechanism of action of famciclovir is as a DNA Polymerase Inhibitor, and DNA Polymerase Inhibitor. |
| 别名 | 泛昔洛韦, BRL 42810 |
| 分子量 | 321.33 |
| 分子式 | C14H19N5O4 |
| CAS No. | 104227-87-4 |
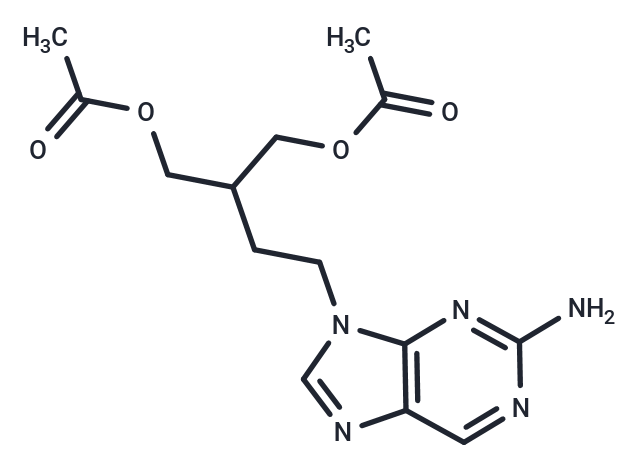
| Smiles | CC(=O)OCC(CCN1C=NC2=C1N=C(N)N=C2)COC(C)=O |
| 密度 | 1.40 g/cm3 (Predicted) |
| 存储 | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. 实际储存温度请以COA为准 | |||||||||||||||||||||||||||||||||||
| 溶解度信息 | Ethanol: 45 mg/mL (140.04 mM), Sonication is recommended. DMSO: 45 mg/mL (140.04 mM), Sonication is recommended. H2O: 59 mg/mL (183.61 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
| 体内实验配方 | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 2 mg/mL (6.22 mM), Sonication is recommended. 请按顺序添加溶剂,在添加下一种溶剂之前,尽可能使溶液澄清。如有必要,可通过加热、超声、涡旋处理进行溶解。工作液建议现配现用。以上配方仅供参考,体内配方并不是绝对的,请根据不同情况进行调整。 | |||||||||||||||||||||||||||||||||||
溶液配制表 | ||||||||||||||||||||||||||||||||||||
Ethanol/DMSO/H2O
该溶液配制表仅适用于固体产品。对于液体产品,请根据标明的浓度或密度计算稀释方案。 | ||||||||||||||||||||||||||||||||||||
对于不同动物的给药剂量换算,您也可以参考 更多