 您的购物车当前为空
您的购物车当前为空
Tenofovir amibufenamide
Synonyms: 艾米替诺福韦, HS-10234Tenofovir amibufenamide (HS-10234) 是一种 Tenofovir 前药,是一种具有口服活性的抗病毒化合物。Tenofovir amibufenamide 对乙型肝炎病毒 (HBV)具有抑制作用,可用于慢性乙型肝炎 (CHB) 研究。
Tenofovir amibufenamide
一键复制产品信息别名 艾米替诺福韦, HS-10234
Tenofovir amibufenamide (HS-10234) 是一种 Tenofovir 前药,是一种具有口服活性的抗病毒化合物。Tenofovir amibufenamide 对乙型肝炎病毒 (HBV)具有抑制作用,可用于慢性乙型肝炎 (CHB) 研究。
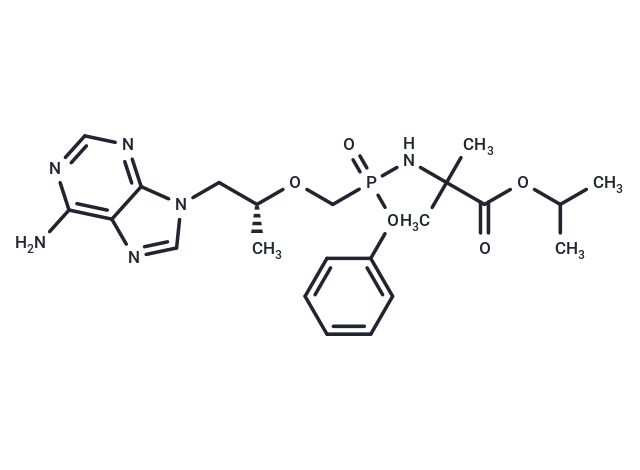
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1 mg | ¥ 2,730 | 现货 | |
| 5 mg | ¥ 4,970 | 现货 | |
| 10 mg | ¥ 6,880 | 现货 | |
| 25 mg | ¥ 9,870 | 现货 | |
| 50 mg | ¥ 13,700 | 现货 | |
| 100 mg | ¥ 18,500 | 现货 |
TargetMol的所有产品仅用作科学研究或药证申报,不能被用于人体,我们不向个人提供产品和服务。请您遵守承诺用途,不得违反法律法规规定用于任何其他用途。
该分子属于定制产品。TargetMol拥有优秀的合成团队,经验和能力,可以为您提供高性价比的产品。 如您有任何问题,欢迎咨询,我们将竭诚为您服务。
凭借在化合物合成方面的丰富经验,我们可以根据您的研究需求为该产品提供快速定制合成服务。
产品介绍
| 产品描述 | Tenofovir amibufenamide (HS-10234) is a Tenofovir prodrug, an antiviral compound with oral activity. Tenofovir amibufenamide inhibits hepatitis B virus (HBV) and can be used in chronic hepatitis B (CHB) studies. |
| 靶点活性 | Anti-HBV:7.29 ± 0.71 nM(EC50) |
| 体外活性 | 在 HBV 阳性的 HepG2.2.15 细胞中,TMF 和 Tenofovir amibufenamide 对 HBV DNA 复制的抑制作用明显强于 TDF。处理 9 天后,TMF 的抗 HBV 活性略强于 Tenofovir amibufenamide(EC50 为 7.29 ± 0.71 nM 对 12.17 ± 0.56 nM)。三种 TFV 酯原药的回调效应依次为 TMF > Tenofovir amibufenamide > TDF。TMF 的这些优势被认为是由于其在临床前动物(SD 大鼠、C57BL/6 小鼠和小猎犬)中更高的生物利用度和更好的靶向负荷,尤其是药理活性代谢物 TFV-DP 的肝脏水平更高,而这与抗 HBV 的疗效密切相关。[1] |
| 体内活性 | 对 Tenofovir amibufenamide 组(25 毫克,96 周)和 TDF 组的安全性进行了全面评估,重点关注骨骼、肾脏和代谢参数。肾脏安全性评估采用非指数估计肾小球滤过率,Tenofovir amibufenamide 组的肾小球滤过率下降幅度小于 TDF 组(P=0.01)。在骨矿物质密度方面,接受 Tenofovir amibufenamide 治疗的患者在第 96 周时脊柱、髋部和股骨颈密度的下降水平明显低于接受 TDF 治疗的患者。此外,第48周后,各组患者的血脂指标均保持稳定,而体重变化仍呈相反趋势。与TDF相比,Tenofovir amibufenamide在第96周时保持了相似的疗效,并且在骨骼和肾脏安全性方面继续保持优势[2]。 |
| 别名 | 艾米替诺福韦, HS-10234 |
| 分子量 | 490.49 |
| 分子式 | C22H31N6O5P |
| CAS No. | 1571076-26-0 |
| Smiles | C([C@H](OCP(OC1=CC=CC=C1)(NC(C(OC(C)C)=O)(C)C)=O)C)N2C=3C(N=C2)=C(N)N=CN3 |
| 存储 | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. 实际储存温度请以COA为准 | |||||||||||||||||||||||||||||||||||
| 溶解度信息 | DMSO: 180 mg/mL (366.98 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
溶液配制表 | ||||||||||||||||||||||||||||||||||||
DMSO
该溶液配制表仅适用于固体产品。对于液体产品,请根据标明的浓度或密度计算稀释方案。 | ||||||||||||||||||||||||||||||||||||
计算器
体内实验配液计算器
剂量转换
对于不同动物的给药剂量换算,您也可以参考 更多




 很棒
很棒
 |
|