购物车
 您的购物车当前为空
您的购物车当前为空
Carbocisteine (Mucodyne) 常被用作具有粘液溶解和祛痰作用的抗感染鼻喷雾剂。
别名 羧甲司坦, S-Carboxymethyl-L-cysteine, Mucodyne, Carbocysteine, Broncodeterge
Carbocisteine (Mucodyne) 常被用作具有粘液溶解和祛痰作用的抗感染鼻喷雾剂。
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 500 mg | ¥ 119 | 现货 | |
| 1 g | ¥ 197 | 现货 | |
| 5 g | ¥ 458 | 现货 |
TargetMol的所有产品仅用作科学研究或药证申报,不能被用于人体,我们不向个人提供产品和服务。请您遵守承诺用途,不得违反法律法规规定用于任何其他用途。

| 产品描述 | Carbocisteine (Mucodyne) is commonly used as an anti-infective nasal spray with mucolytic and expectorant effects. |
| 别名 | 羧甲司坦, S-Carboxymethyl-L-cysteine, Mucodyne, Carbocysteine, Broncodeterge |
| 分子量 | 179.19 |
| 分子式 | C5H9NO4S |
| CAS No. | 25390-17-4 |
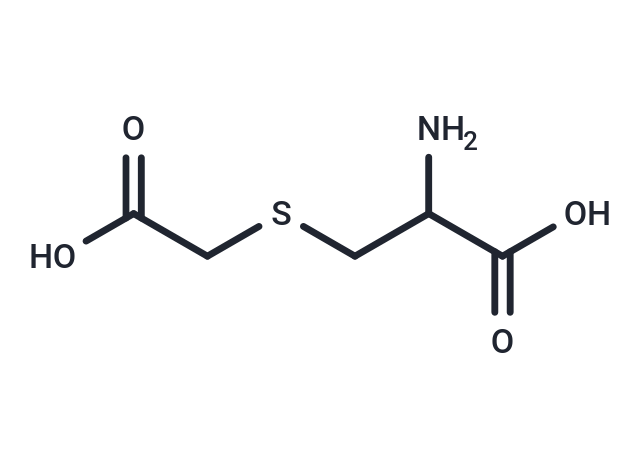
| Smiles | NC(CSCC(O)=O)C(O)=O |
| 密度 | 1.514 g/cm3 (Predicted) |
| 存储 | Keep away from direct sunlight Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. 实际储存温度请以COA为准 | |||||||||||||||
| 溶解度信息 | DMSO: < 1 mg/mL (insoluble or slightly soluble) H2O: 1.71 mg/mL (9.54 mM), Sonication is recommended. | |||||||||||||||
溶液配制表 | ||||||||||||||||
H2O
该溶液配制表仅适用于固体产品。对于液体产品,请根据标明的浓度或密度计算稀释方案。 | ||||||||||||||||
对于不同动物的给药剂量换算,您也可以参考 更多