 您的购物车当前为空
您的购物车当前为空
Vandetanib
一键复制产品信息别名 凡德他尼, ZD6474
Vandetanib (ZD6474) 是一种具有口服活性的VEGFR2/KDR 酪氨酸激酶抑制剂,IC50值为40 nM。它也抑制VEGFR3/FLT4和EGFR/HER1,IC50值分别为110和500 nM。
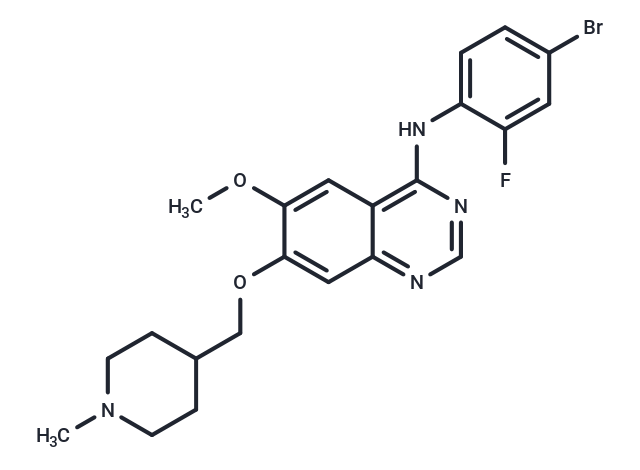
Vandetanib
一键复制产品信息Vandetanib (ZD6474) 是一种具有口服活性的VEGFR2/KDR 酪氨酸激酶抑制剂,IC50值为40 nM。它也抑制VEGFR3/FLT4和EGFR/HER1,IC50值分别为110和500 nM。
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5 mg | ¥ 229 | 现货 | |
| 10 mg | ¥ 372 | 现货 | |
| 25 mg | ¥ 494 | 现货 | |
| 50 mg | ¥ 891 | 现货 | |
| 100 mg | ¥ 1,643 | 现货 | |
| 500 mg | ¥ 3,671 | 现货 | |
| 1 mL x 10 mM (in DMSO) | ¥ 413 | 现货 |
产品介绍
| 产品描述 | Vandetanib (ZD6474) is a potent inhibitor of VEGFR2 (IC50: 40 nM). It also inhibits VEGFR3 and EGFR. |
| 靶点活性 | VEGFR3:110 nM (cell free), VEGFR2:40 nM (cell free), EGFR:500 nM (cell free) |
| 体外活性 | Vandetanib (ZD6474) 是一种强效的KDR/VEGFR2酪氨酸激酶活性抑制剂(IC50:40 nM),对VEGFR3(IC50:110 nM)和EGFR/HER1酪氨酸激酶活性(IC50:500 nM)也有一定抑制作用。ZD6474对KDR酪氨酸激酶的抑制作用能强效抑制VEGF激活的内皮细胞(人脐静脉内皮细胞)增殖(IC50:60 nM)[1]。ZD6474能剂量依赖性抑制小鼠NIH-EGFR成纤维细胞和人MCF-10A ras乳腺癌细胞中EGFR的磷酸化,并在具有功能性EGFR但缺乏VEGFR-2的七种人类细胞系中,剂量依赖性抑制软琼脂生长。ZD6474与紫杉醇或多西他赛联合治疗在体外观察到增长抑制和凋亡的剂量依赖性超加性效应[2]。Vandetanib和neratinib对基础ABCG2-ATP酶活性表现出抑制效果,在相对高浓度(10-20 mM)时,vandetanib能抑制激活的ABCG2-ATP酶活性[3]。 |
| 体内活性 | 给予ZD6474 (2.5 mg/kg, 静脉注射) 能够逆转由VEGF引起的低血压变化(63%),但对由基础成纤维细胞生长因子引起的变化无显著影响。每日一次口服50 mg/kg ZD6474给缺乏胸腺的小鼠(这些小鼠体内已经皮下植入了A549肿瘤细胞)同样显著抑制了由肿瘤引起的新血管生成(5天后抑制率达63%)。对于用50 mg/kg/day ZD6474处理24天的Calu-6肿瘤进行组织学分析显示,非坏死区域内CD31(内皮细胞)染色显著减少(>70%)[1]。ZD6474对携带可触知GEO结肠癌异种移植瘤的裸鼠治疗显示出剂量依赖性的肿瘤生长抑制作用。ZD6474在GEO肿瘤异种移植瘤模型中的抗肿瘤活性,在与紫杉醇联合应用时得到增强。所有使用ZD6474加紫杉醇治疗的小鼠观察到肿瘤退缩,并且伴随着显著增强的抗血管生成抑制作用[2]。Vandetanib (15 mg/kg) 对H1650/PTEN和H1650亲本异种移植瘤的生长具有相似的效果[4]。 |
| 激酶实验 | The ability of ZD6474 to inhibit the kinase activity associated with the VEGFRs KDR, Flt-1, and Flt-4 was determined using a previously described ELISA. Briefly, ZD6474 was incubated with enzyme, 10 mm MnCl2, and 2 μm ATP in 96-well plates coated with a poly(Glu, Ala, Tyr) 6:3:1 random copolymer substrate. Phosphorylated tyrosine was then detected by sequential incubation with a mouse IgG anti-phosphotyrosine 4G10 antibody, a horseradish peroxidase-linked sheep anti-mouse immunoglobulin antibody, and 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid). Microcal Origin software was used to interpolate IC50 values by nonlinear regression. This methodology was adapted to examine selectivity versus tyrosine kinases associated with EGFR, PDGFRβ, Tie-2, FGFR1, c-kit, erbB2, IGF-IR, and FAK. All enzyme assays (tyrosine or serine-threonine) used appropriate ATP concentrations at or just below the respective Km (0.2–14 μm). Selectivity versus serine-threonine kinases (CDK2, AKT, and PDK1) was examined using a relevant scintillation proximity assay (SPA) in 96-well plates. CDK2 assays contained 10 mm MnCl2, 4.5 μm ATP, 0.15 μCi of [γ-33P]ATP/reaction, 50 mm HEPES (pH 7.5), 1 mm DTT, 0.1 mm sodium orthovanadate, 0.1 mm sodium fluoride, 10 mm sodium glycerophosphate, 1 mg/ml BSA fraction V, and a retinoblastoma substrate (part of the retinoblastoma gene, 792–928, expressed in a glutathione S-transferase expression system; 0.22 μm final concentration). Reactions were allowed to proceed at room temperature for 60 min before quenching for 2 h with 150 μl of a solution containing EDTA (62 mm final concentration), 3 μg of a rabbit immunoglobulin anti-glutathione S-transferase antibody and protein A SPA-polyvinyltoluene beads (0.8 mg/reaction). Plates were then sealed, centrifuged (1200 × g for 5 min), and counted on a Topcount NXT Microplate scintillation counter for 30 s [1]. |
| 细胞实验 | HUVEC proliferation in the presence and absence of growth factors was evaluated using [3H]thymidine incorporation. Briefly, HUVECs isolated from umbilical cords were plated (at passage 2–8) in 96-well plates (1000 cells/well) and dosed with ZD6474 ± VEGF or EGF (3 ng/ml) or bFGF (0.3 ng/ml). The cultures were incubated for 4 days (37°C; 7.5% CO2) and then pulsed with 1 μCi/well [3H]thymidine and reincubated for 4 h. Cells were harvested and assayed for the incorporation of tritium using a beta counter. IC50 data were interpolated as described above [1]. |
| 动物实验 | Methodology to enable blood pressure measurement in anesthetized rats was as described previously. Briefly, anesthesia was induced in male Alderley Park rats using α-chloralose by the i.v. route and then maintained with thiopentone via the i.p. route. Once surgical anesthesia was established, the carotid artery was cannulated to enable blood pressure recording using a pressure transducer and a Lectromed MT8P amplifier. The jugular vein was cannulated to allow growth factor administration. Body temperature was maintained with a thermostatically controlled heated blanket coupled to a rectal thermometer. Human VEGF165 (32 μg/kg) or bFGF (40 μg/kg) was administered as a bolus injection [0.1 ml/250 g body weight in 0.85% (w/v) sodium chloride], and a maximal blood pressure drop was recorded within 2 min (typically 26–30 mm Hg). These changes were sustainable for more than 20 min in control experiments. ZD6474 (2.5 mg/kg) or vehicle alone [25% (w/v) hydroxypropyl-β-cyclodextrin in Sorensons phosphate buffer (pH 5.5)] was administered i.v., and blood pressure was recorded 5 min later to determine the effect on growth factor-induced hypotension [1]. |
| 别名 | 凡德他尼, ZD6474 |
| 分子量 | 475.35 |
| 分子式 | C22H24BrFN4O2 |
| CAS No. | 443913-73-3 |
| Smiles | COc1cc2c(Nc3ccc(Br)cc3F)ncnc2cc1OCC1CCN(C)CC1 |
| 密度 | 1.406 |
| 颜色 | White |
| 物理性状 | Solid |
| 存储 | Powder: -20°C for 3 years | In solvent: -80°C for 1 year | Shipping with blue ice/Shipping at ambient temperature. | ||||||||||||||||||||||||||||||
| 溶解度信息 | DMSO: 31.25 mg/mL (65.74 mM), Sonication is recommended. | ||||||||||||||||||||||||||||||
| 体内实验配方 | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 2 mg/mL (4.21 mM), Sonication is recommended. 请按顺序添加溶剂,在添加下一种溶剂之前,尽可能使溶液澄清。如有必要,可通过加热、超声、涡旋处理进行溶解。工作液建议现配现用。以上配方仅供参考,体内配方并不是绝对的,请根据不同情况进行调整。 | ||||||||||||||||||||||||||||||
溶液配制表 | |||||||||||||||||||||||||||||||
DMSO
| |||||||||||||||||||||||||||||||
计算器
体内实验配液计算器
以上为“体内实验配液计算器”的使用方法举例,并不是具体某个化合物的推荐配制方式,请根据您的实验动物和给药方式选择适当的溶解方案。
剂量转换
对于不同动物的给药剂量换算,您也可以参考 更多




 很棒
很棒
 |
|