 您的购物车当前为空
您的购物车当前为空
Deleobuvir sodium
Deleobuvir sodium is the salt form of Deleobuvir, also known as BI207127, a non-nucleoside hepatitis C virus NS5B polymerase inhibitor for the treatment of hepatitis C. Deleobuvir was tested in combination regimens with pegylated interferon and ribavirin, and in interferon-free regimens with other direct-acting antiviral agents including faldaprevir. Deleobuvir showed that a triple combination of deleobuvir, faldaprevir, and ribavirin performed well in HCV genotype 1b patients. Efficacy fell below 50%, however, for dual regimens without ribavirin and for genotype 1a patients. In December 2013, deleobuvir was discontinued since recent findings from phase III trials did not suggest sufficient efficacy.
Deleobuvir sodium
一键复制产品信息Deleobuvir sodium is the salt form of Deleobuvir, also known as BI207127, a non-nucleoside hepatitis C virus NS5B polymerase inhibitor for the treatment of hepatitis C. Deleobuvir was tested in combination regimens with pegylated interferon and ribavirin, and in interferon-free regimens with other direct-acting antiviral agents including faldaprevir. Deleobuvir showed that a triple combination of deleobuvir, faldaprevir, and ribavirin performed well in HCV genotype 1b patients. Efficacy fell below 50%, however, for dual regimens without ribavirin and for genotype 1a patients. In December 2013, deleobuvir was discontinued since recent findings from phase III trials did not suggest sufficient efficacy.
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25 mg | ¥ 15,000 | 8-10周 | |
| 50 mg | ¥ 19,800 | 8-10周 | |
| 100 mg | ¥ 25,500 | 8-10周 |
TargetMol的所有产品仅用作科学研究或药证申报,不能被用于人体,我们不向个人提供产品和服务。请您遵守承诺用途,不得违反法律法规规定用于任何其他用途。
凭借在化合物合成方面的丰富经验,我们可以根据您的研究需求为该产品提供快速定制合成服务。
产品介绍
| 产品描述 | Deleobuvir sodium is the salt form of Deleobuvir, also known as BI207127, a non-nucleoside hepatitis C virus NS5B polymerase inhibitor for the treatment of hepatitis C. Deleobuvir was tested in combination regimens with pegylated interferon and ribavirin, and in interferon-free regimens with other direct-acting antiviral agents including faldaprevir. Deleobuvir showed that a triple combination of deleobuvir, faldaprevir, and ribavirin performed well in HCV genotype 1b patients. Efficacy fell below 50%, however, for dual regimens without ribavirin and for genotype 1a patients. In December 2013, deleobuvir was discontinued since recent findings from phase III trials did not suggest sufficient efficacy. |
| 分子量 | 675.56 |
| 分子式 | C34H32BrN6NaO3 |
| CAS No. | 1370023-80-5 |
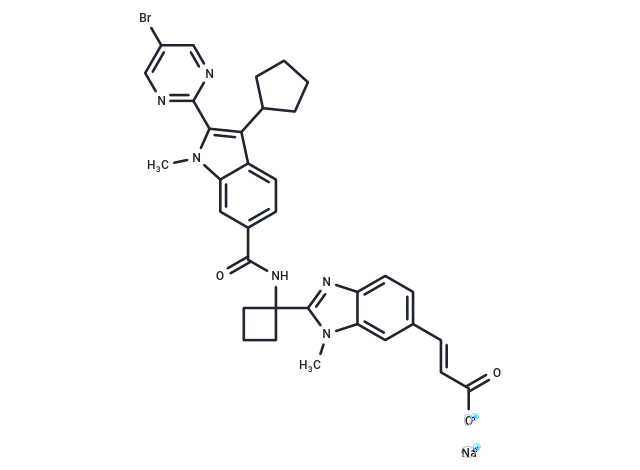
| Smiles | [Na+].Cn1c(c(C2CCCC2)c2ccc(cc12)C(=O)NC1(CCC1)c1nc2ccc(\C=C\C([O-])=O)cc2n1C)-c1ncc(Br)cn1 |
| 存储 | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. 实际储存温度请以COA为准 |
计算器
体内实验配液计算器
剂量转换
对于不同动物的给药剂量换算,您也可以参考 更多





 还可以
还可以
 |
|